Home > Clinical Research Service > CRC Service
Site management / clinical research coordination
Since 2012, Kun Tuo’s CRC team has been successfully providing services in many research sites of different clinical trial projects, in areas such as medical devices, chemical and biological products. Our therapeutic experience includes oncology, cardiovascular, anti-infection, autoimmune diseases, endocrine system disease, neural diseases, respiratory diseases, ophthalmology and digestive diseases. We focus on the following diseases: lung cancer, liver cancer, gastric cancer, Alzheimer’s disease, schizophrenia, epilepsy and diabetes mellitus. We have experience in clinical trials from Phase I to Phase IV, non-interventional outcome studies and medical device studies.
With our CRC team’s specialized support, your projects can initiate earlier than the scheduled timeline and greatly increase enrollment speed – enabling timely and high-quality completion of data entry and significantly improved monitoring efficiency. Our superior CRC services are recognized by sponsors, investigators, study sites and other partner CROs. We have had no major findings at audits/quality control visits.
Our services
Project related training and internal meetingsSite management
- Coordination of site initiation
- Management of research documents and files
- Management of study drugs and supplies
- Management of lab samples and auxiliary examination
- Ethics committee communications and document submission
- Subject recruitment support
- Subject visit support
- Data entry and assisting investigators in resolving queries
- Supporting in safety information reporting and follow-up
- SMV support and other administrative support
- Audit and inspection coordination
- Finance management support
- Site close-out coordination
Our strengths
- Experienced team, efficient management processes
Kun Tuo has a clinical research coordinator (CRC) management team with extensive clinical trial experience. Our managers have years of experience in site management, overseas clinical trials and clinical operations at global pharmaceutical companies. We fully understand the demands of sponsors.
All members of our highly qualified CRC team hold a Bachelor’s or equivalent degree and higher qualifications in nursing, medicine, pharmaceutical sciences or other related medical sciences, with at least a level CET 4 certified English proficiency. Most team members have worked as registered nurses in hospitals and are familiar with the work processes in the hospital environment, as well as the working and communication styles of the investigator team.
Under our project management service model, we develop a project-specific plan which includes risk management, people management and communication planning – all overseen by our CRC project manager. This establishes a firm foundation for providing high-quality services to sponsors.
All of our CRC team members have experience in multi-center international studies and local studies, with extensive work experience in EDC, IVRS, central laboratories and central imaging review. Our highly qualified, experienced team and mature processes allow us to deliver superior service to our customers. - Wide team distribution
Our CRC services are distributed across a number of cities, covering many of the provincial capitals, cities and regions in China where clinical studies are actively conducted. As business needs develop, our service distribution is rapidly extending. The wide geographic allocation of our CRC services not only greatly improves our CRC team’s work efficiency, but also reduces project operating costs on travel. - Our therapeutic expertise
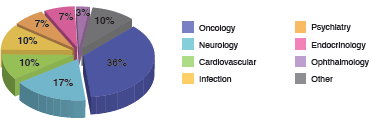
Our flexible CRC services
- Well-developed SOP systems, adapted for clinical studies in China
- Huge clinical research database
- Flexible service model, focused on customer needs
Two CRC service models to choose from
| Overall project management service model | Task-oriented management service model | |
| Team composition | CRC team + CRC PM | Individual CRC or CRC team |
| Timing | At any time from study start-up to site/project closeout | At any time during the project, according to your needs |
| Features | Select CRC service scope according to your needs CRC PM is responsible for project management, communication and coordination | Flexible and cost-effective |

